Health consequences of obesity
This page was started in the framework of an Eduzendium course and needs to be assessed for quality. If this is done, this {{EZnotice}} can be removed.
Introduction
Obesity is a fast growing global epidemic that is rapidly replacing undernutrition and infectious diseases as one of the greatest contributors to worldwide morbidity. Obesity is defined according to Body Mass Index (BMI) and it is generally accepted that a BMI in excess of 30kg/m2 categorises someone as obese. Beyond this point, the risk of morbidity and mortality greatly increases and the main associated health consequences have been summarised in Figure 1. [1]

This article will focus on the cardiovascular (CV) consequences, nonalcoholic fatty liver disease (NAFLD), endocrine changes and psychosocial consequences of obesity as these are generally identified as the most significant complications. The implication of the association between diabetes mellitus type 2 and obesity is of great importance as well, however, since this is such a vast topic it has been discussed separately in the article entitled Diabesity.
Physiological Consequences of Obesity
Cardiovascular Disease in Obesity
Obesity is well known for its association with many serious of diseases, including type 2 diabetes mellitus, coronary heart disease (CHD) amongst many others . BMI has a close correlation with the incidence of several chronic conditions caused by excess fat, and waist circumference correlates with the measure of risk for CHD such as hypertension or blood lipid levels [1].
Obesity increases the risk of cardiovascular disease and premature death which may be indirectly mediated through risk factors associated with the metabolic syndrome . Central deposition of adipose tissue increases the risk of cardiovascular morbidity and mortality, including stroke, congestive heart failure, myocardial infarction and cardiovascular death [3].
Waist-hip ratios are commonly used to assess this type of body fat distribution.
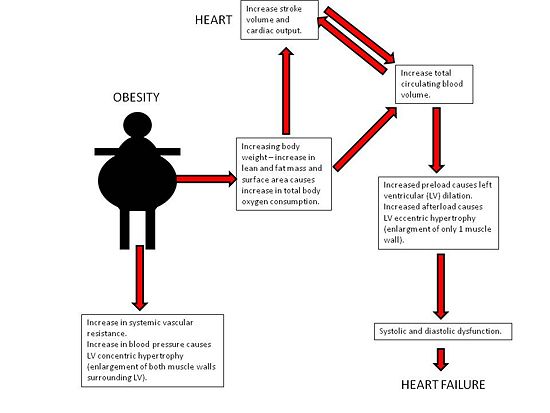
Obesity causes an increase in total body oxygen consumption due to excess lean tissue mass as well as the oxidative demands of metabolically active adipose tissue. This results in an increase in cardiac output [1]. The left ventricle dilates to accommodate the increased venous return with subsequent development of eccentric hypertrophy to keep the wall stress normal[4]. Eventually the ventricle can no longer adapt to volume overload and the dilation of LV results in decreased ventricular contractility[1]. With LV hypertrophy, reduced ventricular compliance alters ability of the chamber to accommodate an increased volume during diastole and this results in diastolic dysfunction. A combination of systolic and diastolic dysfunction leads to clinically significant heart failure [1].
Hypertension also becomes more prevalent with increasing severity of obesity [4]. In men a BMI of <25 or >30 shows a prevalence of hypertension of 15% and 42%, respectively; in women these are 15% and 38%, respectively [4]. Fatal arrhythmias may be the most frequent cause of death among obese people as increased catecholamine and free fatty acid levels may affect repolarization[4]. The Framingham Study shows that sudden cardiac death was 40 times higher in obese men and women. In the NHANES III study, 30% of obese patients with glucose intolerance had a prolonged corrected QT (QTc) interval[4]. A QTc of more than 0.42 seconds was associated with increased mortality in “healthy” obese patients[4]. Schouten et al found that 8% of obese individuals had QTc interval of more than 0.44 seconds and in 2% it was more than 0.46 seconds.
Increased adiposity and reduced physical activity are strong and independent predictors of CHD and death. For each unit increase in BMI, the risk of CHD increases by 8%. However each 1 hour metabolic equivalent increase in activity score decrease CHD risk by 8% [3]. Physical activity attenuates the risks of obesity on coronary health and also increases myocardial oxygen supply, improving myocardial contraction and electrical stability [3]. Various studies have shown that obesity is an independent predictor of coronary artery disease and this is also linked to BMI[4] and that obesity accelerates atherosclerosis many years before the clinical signs become obvious. In autopsies among 15-35 year olds who died from accidental causes, plaques and ulceration in the coronary arteries and abdominal aorta were found and the extent of damage related to the amount of abdominal fat and BMI [4].
The risk of stroke increases with increased BMI and waist-hip ratio. In the prospective Physician’s Health study, results showed that an increase of 1 BMI unit, increased the rate of ischemic stroke by 4% and 6% for haemorrhagic stroke. The underlying mechanisms linking increased BMI to increased stroke risk are not clear but it is thought that it could be mediated by the prothrombotic and proinflammatory state in obesity[4]. Adipose tissue is considered as an active endocrine organ[3]. Release of adipokines (e.g. leptin and adiponectin), proinflammatory cytokines (IL-6 and CRP) and hypofibrinolytic factors (PAI-1) might, together, lead to increased oxidative stress and endothelial dysfunction, finally promoting atherosclerosis which then leads to stroke [3]. Terao et al (2008) investigated the effect of inflammatory and injury reposonse to ischaemic stroke in obese mice, and discovered that when the middle cerebral artery (MCA) was occluded and reperfused, the inflammatory and injury responses were worse in obese mice (ob/ob) than in wild type mice[5]. Monoctye chemoattractant protein-1 appears to be involved in the exaggerated responses to ischaemic stroke in obese mice.
This page was started in the framework of an Eduzendium course and needs to be assessed for quality. If this is done, this {{EZnotice}} can be removed.
Introduction
Obesity is a fast growing global epidemic that is rapidly replacing undernutrition and infectious diseases as one of the greatest contributors to worldwide morbidity. Obesity is defined according to Body Mass Index (BMI) and it is generally accepted that a BMI in excess of 30kg/m2 categorises someone as obese. Beyond this point, the risk of morbidity and mortality greatly increases and the main associated health consequences have been summarised in Figure 1. [1]

This article will focus on the cardiovascular (CV) consequences, nonalcoholic fatty liver disease (NAFLD), endocrine changes and psychosocial consequences of obesity as these are generally identified as the most significant complications. The implication of the association between diabetes mellitus type 2 and obesity is of great importance as well, however, since this is such a vast topic it has been discussed separately in the article entitled Diabesity.
Physiological Consequences of Obesity
Cardiovascular Disease in Obesity
Obesity is well known for its association with many serious of diseases, including type 2 diabetes mellitus, coronary heart disease (CHD) amongst many others . BMI has a close correlation with the incidence of several chronic conditions caused by excess fat, and waist circumference correlates with the measure of risk for CHD such as hypertension or blood lipid levels [1].
Obesity increases the risk of cardiovascular disease and premature death which may be indirectly mediated through risk factors associated with the metabolic syndrome . Central deposition of adipose tissue increases the risk of cardiovascular morbidity and mortality, including stroke, congestive heart failure, myocardial infarction and cardiovascular death [3].
Waist-hip ratios are commonly used to assess this type of body fat distribution.
Obesity causes an increase in total body oxygen consumption due to excess lean tissue mass as well as the oxidative demands of metabolically active adipose tissue. This results in an increase in cardiac output [1]. The left ventricle dilates to accommodate the increased venous return with subsequent development of eccentric hypertrophy to keep the wall stress normal[4]. Eventually the ventricle can no longer adapt to volume overload and the dilation of LV results in decreased ventricular contractility[1]. With LV hypertrophy, reduced ventricular compliance alters ability of the chamber to accommodate an increased volume during diastole and this results in diastolic dysfunction. A combination of systolic and diastolic dysfunction leads to clinically significant heart failure [1].
Hypertension also becomes more prevalent with increasing severity of obesity [4]. In men a BMI of <25 or >30 shows a prevalence of hypertension of 15% and 42%, respectively; in women these are 15% and 38%, respectively [4]. Fatal arrhythmias may be the most frequent cause of death among obese people as increased catecholamine and free fatty acid levels may affect repolarization[4]. The Framingham Study shows that sudden cardiac death was 40 times higher in obese men and women. In the NHANES III study, 30% of obese patients with glucose intolerance had a prolonged corrected QT (QTc) interval[4]. A QTc of more than 0.42 seconds was associated with increased mortality in “healthy” obese patients[4]. Schouten et al found that 8% of obese individuals had QTc interval of more than 0.44 seconds and in 2% it was more than 0.46 seconds.
Increased adiposity and reduced physical activity are strong and independent predictors of CHD and death. For each unit increase in BMI, the risk of CHD increases by 8%. However each 1 hour metabolic equivalent increase in activity score decrease CHD risk by 8% [3]. Physical activity attenuates the risks of obesity on coronary health and also increases myocardial oxygen supply, improving myocardial contraction and electrical stability [3]. Various studies have shown that obesity is an independent predictor of coronary artery disease and this is also linked to BMI[4] and that obesity accelerates atherosclerosis many years before the clinical signs become obvious. In autopsies among 15-35 year olds who died from accidental causes, plaques and ulceration in the coronary arteries and abdominal aorta were found and the extent of damage related to the amount of abdominal fat and BMI [4].
The risk of stroke increases with increased BMI and waist-hip ratio. In the prospective Physician’s Health study, results showed that an increase of 1 BMI unit, increased the rate of ischemic stroke by 4% and 6% for haemorrhagic stroke. The underlying mechanisms linking increased BMI to increased stroke risk are not clear but it is thought that it could be mediated by the prothrombotic and proinflammatory state in obesity[4]. Adipose tissue is considered as an active endocrine organ[3]. Release of adipokines (e.g. leptin and adiponectin), proinflammatory cytokines (IL-6 and CRP) and hypofibrinolytic factors (PAI-1) might, together, lead to increased oxidative stress and endothelial dysfunction, finally promoting atherosclerosis which then leads to stroke [3]. Terao et al (2008) investigated the effect of inflammatory and injury reposonse to ischaemic stroke in obese mice, and discovered that when the middle cerebral artery (MCA) was occluded and reperfused, the inflammatory and injury responses were worse in obese mice (ob/ob) than in wild type mice[5]. Monoctye chemoattractant protein-1 appears to be involved in the exaggerated responses to ischaemic stroke in obese mice.
Non-alcoholic liver disease and obesity
Definition: Nonalcoholic fatty liver disease can be defined as an accumulation of fat, mainly triglycerides in the hepatocytes that exceeds 5% of the liver weight, this is a condition known as steatosis [7]. If left untreated steatosis can lead to the development of steatohepatitis, which can result in fibrosis, cirrhosis and eventual liver failure. NAFLD therefore refers to a wide range of liver damage [8]
Clinical presentation: Patients presenting with NAFLD complain of fatigue, malaise and feelings of discomfort or “fullness” in the upper right abdomen. Laboratory tests reveal a mild to moderate increase in serum levels of alanine aminotransferase, aspartate aminotransferase or both [8]. The ratio of aspartate aminotransferase to alanine aminotransferase is usually less than 1, however as the degree of fibrosis in the liver advances, this ratio increases [7].
Risk Factors: Insulin resistance and therefore NAFLD are most commonly associated with the central obesity phenotype. Visceral fat is found to have a greater lipolytic potential than subcutaneous fat. The fatty acids which are released from visceral fat during lipolysis drain straight into the portal circulation. It is the increased lecels of free fatty acids in the circulation which are thought to be responsible for insulin resistance [9].
Prevalence: NAFLD is known to affect 10 -24% of the world’s population, however this prevalence increases to 57.5% to 74% in obese persons. Simularly NAFLD is thought to affect 2.6% of children a statistic which rises to 22.5% to 52.8% of obese children [8].
The role of Insulin Resistance in NAFLD: Insulin resistance, a condition seen in conjunction with obesity and type 2 diabetes mellitus is thought to be the leading cause of NAFLD. It is thought that fat accumulation in the hepatocytes occurs via two distinct mechanisms, namely lipolysis and hyperinsulinaemia. Microsomal ω-oxidation has been found to produce clinically significant amounts of cytotoxic dicarboxdylic acids. This pathway of fatty acid metabolism is closely related to mitochondrial β-oxidation and peroxisomal β-oxidation. A lack in the enzymes associated with peroxisomal β-has been identified as a major cause of steatosis and steatohepatitis. An example of this is deficiency of acyl-coenzyme A oxidase which disrupts disrupt the oxidation of long fatty acid chains and dicarboxilic acids, which in turn leads to microvesicular steatosis and steatohepatitis. Loss of function of acyl-coenzyme A oxidase has also causes sustained hyperactivation of peroxisome proliferator activated receptor-α (PPAR- α), leading to upregulation of PPAR- α regulated genes [10]. Studies have also been conducted which suggest that PPAR- α is responsible for the promoting the synthesis of uncoupling protein 2 in the liver [11]. Increased levels of fatty acids in the liver provide a source of oxidative stress, which in turn is thought to be responsible for the progression from steatosis to steatohepatitis and finally to cirrhosis.the reactive oxygen species is found within the mitochondria of the cell and is thought to trigger steatohepatitis and fibrosis via three different mechanisms, lipid peroxidation, cytokine induction and induction of FAS ligand [8].
The role of the Keratin cytosleleton in NAFLD: Mature and differentiated hepatocytes are the epithelial cells of the liver and normally express the type I keratin keratin 8 anad the type II keratin keratin 18, these are arranged into intermediate filaments in the cytoplasmof the cell. During diseases such as NAFLD disruption to the keratin cytoskeleton is routinely observed in hepatocytes. NAFLD is characterised by steatosis, hepatocyte ballooning, cytoplasmic inclusions such as Mallory bodies, pericellular fibrosis and inflammation [12]. Observation of ballooned hepatocytes reveals reduced density and in some cases loss of intermediate filaments, and the Mallory bodies are seen to exist of misfolded and aggregated keratins [12]. A study carried out by ….???.... has revealed that disruption of the cytoskeleton is due to oxidative stress. Mallory bodies and ballooned hepatocytes were recrested in hepatocytes of mice by chronic intoxication with Griseofulvin or 3,5-diethoxycarbonyl-1,4 dihydrocollidine [13]. These two reactants were metabolised by cytochrome P450, which in return lead to the formation of methyl radicals [14]. It is assumed that the mechanism of oxidative injury seen in the study was the same as that which occurs in NAFLD, but in the case of the disease fatty acids are responsible for the production of reactive oxygen species [12].
In conclusion NAFLD is a disease found to be closely linked to both central obesity and insulin resistance which affects a large number of the world’s population
Endocrine Changes in Obesity
It is well documented that obesity is associated with changes in the normal endocrine profile. Many studies have focused particularly on the alterations in the sex steroid profile of obese individuals. This section of the article will briefly discuss the types of changes observed and their consequences on the health of the individual.
Oestrogens: Oestrogens are synthesized by aromatization of circulating testosterones, catalysed by the enzyme aromatase [15]. This process occurs at many sites throughout the body including adipose tissue [16], [17]. Therefore, an increase in adipose tissue mass, results in a greater capacity for aromatization, and an increase in oestrogen levels [18].
In premenopausal, non-pregnant women, the principal site of aromatization is the ovaries [15]. and only a minor proportion of oestrogen comes from adipose tissue. However, in postmenoapusal women, peripheral aromatisation is enhanced and adipose tissue becomes the main site of oestrogen production [15].
Androgens: The increased capacity for aromatization results in hypoandrogenism in males, as a higher proportion of circulating testosterone is converted to oestrogen [19]. Other factors contributing to the decrease in circulating testoerone include insulin resistance and the suppression of the hypothalamic pituitary testicular axis [19].
In contrast, in obese premenopausal women, obesity is associated with an increase in free testosterone levels [20] This is thought to be partly mediated by the increased levels of insulin and IGF-1 associated with obesity [20]15.) Furthermore, as is the case for oestrogen, adipose tissue is an important site of peripheral testosterone production, due to local expression of 17 beta hydroxysteroid dehydrogenase [20]. Therefore there is a positive association between adipose tissue mass and androgen concentration.
SHBG:Sex steroids are highly lipophilic and are therefore carried in the circulation bound to proteins – sex hormone binding globulins (SHBG.) It is well documented that obesity results in a decreased concentration of SHBG (16l.) This is thought to be associated with the rise in insulin levels associated with obesity, as insulin inhibits hepatic synthesis of SHBG (16.) The decreased concentration of binding protein results in an increase in the free fraction of sex steroids [20].
| Female Changes | Male Changes | |
|---|---|---|
| Oestrogen | Increase | Increase |
| Androgens | Increase | Decrease |
| SHBG | Decrease | Decrease |
Fig 2: Summary of the endocrine changes in obesity
Obesity and Cancer
It is estimated that 10% of all cancer deaths among non-smokers are related to obesity (17.) Many hypotheses have been proposed to explain this. It is hypothesised that alterations in endogenous hormone metabolism mediate the effects of obesity on cancer risk, due to sex steroid hormone regulation of cell proliferation, differentiation and apoptosis [17].
Many types of cancer are more prevalent in obese subjects, the most widely studied of which are breast and endometrial cancer. These types of cancer are associated with an increase in oestrogen concentration, a decrease in plasma SHBG and an increase in androgen levels. [17], 14) These observations have led to the establishment of the unopposed oestrogen hypothesis, which suggests that mitotic activity of cells is increased in the presence of oestrogen, unopposed by progestogens (18.) Increased mitotic activity leads to a higher prevalence of mutations occurring, thus increasing the risk of developing cancer.
Obesity and Infertility
Obesity is thought to account for around 6% of primary infertility (17.)
Women:Many studies have observed increased risk of anovulatory infertility in obese women (15.) This is thought to be due to hyperandrogenism in which high androgen levels increase apoptosis of the granulosa cells, as well as damaging the endometrium and developing oocytes [18]., 15). Excess oestrogen also contributes to infertility by reducing gonadotrophin secretion through excess negative feedback (17, [19].
One of the commonest reproductive disorders in women is PCOS, which affects 5-10% of women of reproductive age (19.) This syndrome is characterized by anovulatory infertility, obesity, hirsutism, multiple ovarian cysts and insulin resistance (16.) Despite their being a well established association between obesity and PCOS, it remains unknown which is the cause and which is the effect. (15, 19) Figure 2 summarises the proposed endocrine basis of this syndrome.
Men:"Hyperestrogenic hypogonadotropic hypogonadism" in obese men results in high oestrogen levels, low testosterone levels and subsequent subfertility [19].
Many studies have reported a negative association between spermatogenesis and increasing BMI [19]. The mechanism mediating this association is yet to be identified. However, it has been suggested that hypoandrogenism in obese males may result in a reduced concentration of testosterone within the testes [19]. The observed hyperestrogenism may also contribute to infertility as it causes inappropriate suppression of the hypothalamic-pituitary-gonadal axis, resulting in reduced spermatogenesis. Obesity has also been associated with erectile dysfunction [19].
In summary, alterations in sex steroid profiles as a result of obesity have many adverse effects on health. The most well documented health consequences are cancer and infertility.
Psychosocial Consequences of Obesity
The relationship between obesity and mental health has been the subject of continuous debate over the past 30 years and remains a topic of extensive deliberation [21]. Early studies exploring the relationship were consistent with the “jolly fat” hypothesis, suggesting that obesity confers a protective role against anxiety and depressive disorders;1 however, the weight of more recent studies contradict the “jolly fat” hypothesis and suggest that the increasing global prevalence of both depression and obesity are functionally linked. [22] 2,3
Recent hypotheses linking obesity and aggression: The underlying mechanisms and direction of an obesity-depression link remain largely unknown,3 although recent research has indicated that gender, obesity severity, comorbid physical illness, stress and abdominal fat distribution are important mediating risk factors for the development of an obesity-mental disorder link.1,2,4,5, These newly discovered mediators give rise to new hypotheses, involving over-activity of the hypothalamic-pituitary-adrenocortical axis,1,2 side-effects of medication for depression, and the social stigmatization of obesity.2
Experimental evidence: A recent nationally representative Canadian study, whose methodology controlled for sociodemographic factors and comorbid physical health problems, found significant positive relationships between obesity and an array of lifetime psychiatric disorders and past-year mood and anxiety disorders.2 These conclusions are consistent with current literature.6,5,7,8 Further subgroup analyses revealed that obese women had a greater susceptibility to specific mental disorders compared to men, including depression, mania, panic attacks, panic disorder, social phobia and agoraphobia (with and without panic).3 These findings are consistent with previous research, and thus reinforce the notion that obese women have an enhanced vulnerability towards mental disorders.2,4,5,7,9 This result may be explained in terms of the increased consciousness amongst women to conform to a socially desirable image and weight.3 The study also positively linked obesity to suicidal behaviours and negatively linked obesity with past-year drug dependence3; both these findings are supportive of existing literature.6,7,10 Mather et al. (2009) suggest that the former may be attributed to the social stigmatization attached to obesity, inducing feelings of decreased self-worth and decreased self-esteem that fuel suicidal thoughts; and the latter due to protective effects of obesity, arising through food and addictive drugs competing for the same reward sites in the brain.3,11
Contradictory experimental evidence: At face value, this data appears convincing, however, it is important to note that some studies fail to identify mental disorders as a psychosocial consequence of obesity.12,13 Similarly, whilst much research is indicative of sex differences between obesity and mental disorders,1,5 this is not a unanimous conclusion.6,7,14 In addition, some research only identifies associations amongst the severely obese, who illustrate a BMI of >35kg/m2.5 These variable conclusions may in part be attributed to methodological differences between studies.5
Conclusion
Obesity is important as a cause and an effect of many different health problems. Therefore the current obesity epidemic is putting considerable strain on various worldwide healthcare systems. Gaining a clear understanding of the cause of the obesity epidemic and the pathogenesis of the associated health consequences is vital in finding appropriate management and treatment strategies.
References
1. Kopelman P.G. Obesity as a medical problem. Nature 2000; 404:635-642.
2. Boon NA, Colledge NR, Walker BR. Davidson's Principles and Practice of Medicine. 2006. p 111
3. Gaal L.F.V. et al. Mechanisms linking obesity with cardiovascular disease. Nature 2006; 444:875-879.
4. Mathew B. et al. Obesity: effects on cardiovascular disease and its diagnosis. J Am Board Fam Me 2008; 21:562-568.
5. Terao S. et al. Inflammatory and injury responses to ischemic stroke in obese mice. Stroke; 2008. 39:943-950
6. Paul Angulo, Obesity and Nonalcoholic fatty Liver Disease, Nutrition reviews, vol 65, 6, 2007, s57-s63
7. Paul Angulo, Nonalcoholic fatty liver disease, New England Journal of Medicine, vol346, 16, 2002, 1221-1231
8. Kurt Zatloukal, conny Stumptner, Andrea Fuchsbichler, Peter Fickert, Carolin Lackner, Michael Trauner and Helmut Denk, The Keratin cytoskeleton in liver diseases, journal of pathology, 2003 vol 204: 367-376
9. Stranges S, Dorn JM, Multi P, et al. Body fat distribution, relative weight, and liver enzyme levels: a population based study. Hepatology. 2004; 39; 754-763
10. Fan Cy, Pan J, Usuda N, Yelandi AV, Rao MS, Reddy JK, Steatohepatitis, spontaneousperoxisome proliferation and liver tumours in mice lacking fatty acyl CoA ocidase; Implications fotedr peroxisome proliferator activated receptor alpha ligand metabolism. J Bio Chem 1998;273;15639-15645
11. Chavin KD, Yang SQ, Lin NZ et al. Obesity increases expression of uncoupling protein 2 in hepatocytes and promotes liver ATP depletion. J biol chem 1999;274:5692-5700
12. Denk H, Gschnait F, Wolff K. Hepatocellular hyalin (Mallory bodies) in long term griseofulvin-treated mice: a new experimental model for the study of hyalin formation. Lab invest 1975; 32:773-776
13. Telphy Tr, coffman BL, Ingall G et al, identification of N-methylprotophorphyran ix in livers of untreated mice and mice treated with 3,5- dieethoxycarbonyl-1.4-dihydorcollidine; source of the methyl group; Arch Bio Chem Biophys 1981; 212; 120-126
14. Purohit A, Reed MJ. Regulation of oestrogen synthesis in postmenopausal women. Steroids 2002: 67; 979-983 (9)
15. Bianchini F, Kaaka R, Vainio H. Overweight, obesity, and cancer risk. The Lancet Oncology 2002; 3: 565-574 (10)
16, Bates GW, Whitworth N. Effects of obesity on sex steroid metabolism. Journal Chronic Diseases 1982; 35: 893 (11)
17. Simpson ER. Role of aromatase in sex steroid action. Journal of Molecular Endocrinology (2000);25: 149-156 (12)
18. Hammoud AO, Gibson M, Peterson CM et al. Obesity and Male Reproductive Potential. Journal of Andrology: 27; 619 – 625 (13)
19. Lukanova A, Lundin E et al. Body mass index, circulating levels of sex steroid hormones, IGF-1 and IGF-binding protein-3: a cross-sectional study in healthy women. European Journal of Enodcrinology 2004; 190: 161-171 (14)
20. Metwally M, Li TC, Ledger L. The impact of obesity on female reproductive function. Obesity Reviews 2007; 8: 515-523 (15)
21. Bray AG. Obesity and Reproduction. Human Reproduction 1997; 12: 26-32 (16)
22. Haslam, D.W. and James, W.P Obesity. Lancet 2995; 366:1197-1209. (17)
23. Key TJ, Pike MC. The dose-effect relationship between ‘unopposed’ oestrogens and endometrial mitotic rate: its central role in explaining and predicting endometrial cancer risk. British Journal of Cancer 1988: 57; 205-12 (18)
24. Strauss JF, Dunaif A. Molecular Mysteries of Polycystic Ovary Syndrome. Molecular Endrocinology 1999: 13; 800-805 (19)
25. Rivenes, A.C., Harvey, S.B., and Mykltun, A. (2009). The relationship between abdominal fat, obesity, and common mental disorders: Results from the HUNT Study. Journal of Psychosomatic Research, 66: 269-275. (R1)
26. Stunkard, A.J., Faith, M.S. and Allison, K.C. (2003). Depression and Obesity. Society of Biological Psychiatry, 54: 330-337. (R2)
27. Mather, A.A., Brian, J.C., Enns, M.W. and Sareen, J. (2009). Association of obesity with psychiatric disorders and suicidal behaviours in a nationally representative sample. Journal of Psychosomatic Research, 66: 277-285. (R3)
28. Simon, G.E., Ludman, E.J., Linde, J.A., Operskalski, B.H., Ichikawa, L., Rohde, P., Finch, E.A. and Jeffery, R.W. (2008). Association between obesity and depression in middle-aged women. Gen Hosp Psychiatry, 30(1): 32-39. (R4)
29. Scott, K.M., Bruffaerts, R., Simon, G.E., Alonso, J., Angermeyer, M., de Girolamo, G., Demyttenaere, K., Gasquet, I., Haro, J.M., Karam, E., Kessler, R.C., Levinson, D., Mora, M.E.M., Browne, M.O., Ormel, J.H., Villa, J.P., Uda, H. and von Korff, M. (2008). Obesity and Mental Disorders in the General Population: Results from the World Mental Health Surveys. Int J Obes, 32(1):192-200. (R5)
30, Simon, G.E., Von Korff, M., Saunders, K., Miglioretti, D.L., Crane, P.K., van Belle, G. and Kessler, R.C. (2006). Association Between Obesity and Psychiatric Disorders in the US Adult Population. Arch Gen Psychiatry, 63: 824-830. (R6)
31. Carpenter, K.M., Hasin, D.S., Allison, D.B. and Faith, M.S. (2000) Relationships between obesity and DSM-IV major depressive disorder, suicide ideation, and suicide attempts: results from a general population study. Am J Public Health, 90:251–257. (R7)
32. Onyike, C.U., Crum R.M. and Lee H.B. (2003). Is obesity associated with major depression? Results from the third National Health and Nutrition Examination Survey. Am J Epidemiol, 158:1139–1147. (R8)
33. Scott, K.M., Oakley Browne, M.A., McGee, M.A. and Wells, J.E. (2006). Mental-physical comorbidity in Te Rau Hinengaro: the New Zealand Mental Health Survey (NZMHS). Australian and New Zealand Journal of Psychiatry, 40:882–888. (R9)
34. Dong, C., Li, W.D. and Li, D. (2006). Extreme obesity is associated with suicide attempts: results from a family study. Int J Obes, 30: 388-390. (R10)
35. Kleiner, K.D., Gold, M.S., Frost-Pineda, K. (2004). Body mass index and alcohol use. J Addict Dis, 23:105-118. (R11)
36. Hasler, G., Pine, D.S. and Gamma, A. (2004). The associations between psychopathology and being overweight: A 20-year prospective study. Psychol Med, 34:1047–157. (R12)
37. Faith, M.S., Matz, P.E. and Jorge, M.A. (2002). Obesity-depression associations in the population. J Psychosom Res, 53:935–942. (R13)
38. Onyike, C.U., Crum, R.M., Lee, H.B., Lyketsos, C.G. and Eaton, W.W. (2003). Is obesity associated with major depression? Results from the Third National Health and Nutrition Examination Survey. American Journal of Epidemiology. 158(12):1139–1147. (R14)
Endocrine Changes in Obesity
It is well documented that obesity is associated with changes in the normal endocrine profile. Many studies have focused particularly on the alterations in the sex steroid profile of obese individuals. This section of the article will briefly discuss the types of changes observed and their consequences on the health of the individual.
Oestrogens: Oestrogens are synthesized by aromatization of circulating testosterones, catalysed by the enzyme aromatase (12.) This process occurs at many sites throughout the body including adipose tissue (9, 10). Therefore, an increase in adipose tissue mass, results in a greater capacity for aromatization, and an increase in oestrogen levels (11.)
In premenopausal, non-pregnant women, the principal site of aromatization is the ovaries (12) and only a minor proportion of oestrogen comes from adipose tissue. However, in postmenoapusal women, peripheral aromatisation is enhanced and adipose tissue becomes the main site of oestrogen production (12.)
Androgens: The increased capacity for aromatization results in hypoandrogenism in males, as a higher proportion of circulating testosterone is converted to oestrogen (13.) Other factors contributing to the decrease in circulating testoerone include insulin resistance and the suppression of the hypothalamic pituitary testicular axis (13.)
In contrast, in obese premenopausal women, obesity is associated with an increase in free testosterone levels (14.) This is thought to be partly mediated by the increased levels of insulin and IGF-1 associated with obesity (14, 15.) Furthermore, as is the case for oestrogen, adipose tissue is an important site of peripheral testosterone production, due to local expression of 17 beta hydroxysteroid dehydrogenase (14.) Therefore there is a positive association between adipose tissue mass and androgen concentration.
SHBG:Sex steroids are highly lipophilic and are therefore carried in the circulation bound to proteins – sex hormone binding globulins (SHBG.) It is well documented that obesity results in a decreased concentration of SHBG (16l.) This is thought to be associated with the rise in insulin levels associated with obesity, as insulin inhibits hepatic synthesis of SHBG (16.) The decreased concentration of binding protein results in an increase in the free fraction of sex steroids (14.)
| Female Changes | Male Changes | |
|---|---|---|
| Oestrogen | Increase | Increase |
| Androgens | Increase | Decrease |
| SHBG | Decrease | Decrease |
Fig 2: Summary of the endocrine changes in obesity
Obesity and Cancer
It is estimated that 10% of all cancer deaths among non-smokers are related to obesity (17.) Many hypotheses have been proposed to explain this. It is hypothesised that alterations in endogenous hormone metabolism mediate the effects of obesity on cancer risk, due to sex steroid hormone regulation of cell proliferation, differentiation and apoptosis (10.)
Many types of cancer are more prevalent in obese subjects, the most widely studied of which are breast and endometrial cancer. These types of cancer are associated with an increase in oestrogen concentration, a decrease in plasma SHBG and an increase in androgen levels. (10, 14) These observations have led to the establishment of the unopposed oestrogen hypothesis, which suggests that mitotic activity of cells is increased in the presence of oestrogen, unopposed by progestogens (18.) Increased mitotic activity leads to a higher prevalence of mutations occurring, thus increasing the risk of developing cancer.
Obesity and Infertility
Obesity is thought to account for around 6% of primary infertility (17.)
Women:Many studies have observed increased risk of anovulatory infertility in obese women (15.) This is thought to be due to hyperandrogenism in which high androgen levels increase apoptosis of the granulosa cells, as well as damaging the endometrium and developing oocytes (11, 15). Excess oestrogen also contributes to infertility by reducing gonadotrophin secretion through excess negative feedback (17, 13.)
One of the commonest reproductive disorders in women is PCOS, which affects 5-10% of women of reproductive age (19.) This syndrome is characterized by anovulatory infertility, obesity, hirsutism, multiple ovarian cysts and insulin resistance (16.) Despite their being a well established association between obesity and PCOS, it remains unknown which is the cause and which is the effect. (15, 19) Figure 2 summarises the proposed endocrine basis of this syndrome.
Men:"Hyperestrogenic hypogonadotropic hypogonadism" in obese men results in high oestrogen levels, low testosterone levels and subsequent subfertility (13.)
Many studies have reported a negative association between spermatogenesis and increasing BMI (13.) The mechanism mediating this association is yet to be identified. However, it has been suggested that hypoandrogenism in obese males may result in a reduced concentration of testosterone within the testes (13.) The observed hyperestrogenism may also contribute to infertility as it causes inappropriate suppression of the hypothalamic-pituitary-gonadal axis, resulting in reduced spermatogenesis. Obesity has also been associated with erectile dysfunction (13.)
In summary, alterations in sex steroid profiles as a result of obesity have many adverse effects on health. The most well documented health consequences are cancer and infertility.
Psychosocial Consequences of Obesity
The relationship between obesity and mental health has been the subject of continuous debate over the past 30 years and remains a topic of extensive deliberation [23]. Early studies exploring the relationship were consistent with the “jolly fat” hypothesis, suggesting that obesity confers a protective role against anxiety and depressive disorders;1 however, the weight of more recent studies contradict the “jolly fat” hypothesis and suggest that the increasing global prevalence of both depression and obesity are functionally linked. [24] 2,3
Recent hypotheses linking obesity and aggression: The underlying mechanisms and direction of an obesity-depression link remain largely unknown,3 although recent research has indicated that gender, obesity severity, comorbid physical illness, stress and abdominal fat distribution are important mediating risk factors for the development of an obesity-mental disorder link.1,2,4,5, These newly discovered mediators give rise to new hypotheses, involving over-activity of the hypothalamic-pituitary-adrenocortical axis,1,2 side-effects of medication for depression, and the social stigmatization of obesity.2
Experimental evidence: A recent nationally representative Canadian study, whose methodology controlled for sociodemographic factors and comorbid physical health problems, found significant positive relationships between obesity and an array of lifetime psychiatric disorders and past-year mood and anxiety disorders.2 These conclusions are consistent with current literature.6,5,7,8 Further subgroup analyses revealed that obese women had a greater susceptibility to specific mental disorders compared to men, including depression, mania, panic attacks, panic disorder, social phobia and agoraphobia (with and without panic).3 These findings are consistent with previous research, and thus reinforce the notion that obese women have an enhanced vulnerability towards mental disorders.2,4,5,7,9 This result may be explained in terms of the increased consciousness amongst women to conform to a socially desirable image and weight.3 The study also positively linked obesity to suicidal behaviours and negatively linked obesity with past-year drug dependence3; both these findings are supportive of existing literature.6,7,10 Mather et al. (2009) suggest that the former may be attributed to the social stigmatization attached to obesity, inducing feelings of decreased self-worth and decreased self-esteem that fuel suicidal thoughts; and the latter due to protective effects of obesity, arising through food and addictive drugs competing for the same reward sites in the brain.3,11
Contradictory experimental evidence: At face value, this data appears convincing, however, it is important to note that some studies fail to identify mental disorders as a psychosocial consequence of obesity.12,13 Similarly, whilst much research is indicative of sex differences between obesity and mental disorders,1,5 this is not a unanimous conclusion.6,7,14 In addition, some research only identifies associations amongst the severely obese, who illustrate a BMI of >35kg/m2.5 These variable conclusions may in part be attributed to methodological differences between studies.5
Conclusion
Obesity is important as a cause and an effect of many different health problems. Therefore the current obesity epidemic is putting considerable strain on various worldwide healthcare systems. Gaining a clear understanding of the cause of the obesity epidemic and the pathogenesis of the associated health consequences is vital in finding appropriate management and treatment strategies.
References
1. Kopelman P.G. Obesity as a medical problem. Nature 2000; 404:635-642. (2)
2. Boon NA, Colledge NR, Walker BR. Davidson's Principles and Practice of Medicine. 2006. p 111 (1)
3. Gaal L.F.V. et al. Mechanisms linking obesity with cardiovascular disease. Nature 2006; 444:875-879. (3)
4. Mathew B. et al. Obesity: effects on cardiovascular disease and its diagnosis. J Am Board Fam Me 2008; 21:562-568. (4)
5. Terao S. et al. Inflammatory and injury responses to ischemic stroke in obese mice. Stroke; 2008. 39:943-950 (5)
6. Paul Angulo, Obesity and Nonalcoholic fatty Liver Disease, Nutrition reviews, vol 65, 6, 2007, s57-s63 (6)
7. Paul Angulo, Nonalcoholic fatty liver disease, New England Journal of Medicine, vol346, 16, 2002, 1221-1231 (7)
8. Kurt Zatloukal, conny Stumptner, Andrea Fuchsbichler, Peter Fickert, Carolin Lackner, Michael Trauner and Helmut Denk, The Keratin cytoskeleton in liver diseases, journal of pathology, 2003 vol 204: 367-376 (8)
9. Stranges S, Dorn JM, Multi P, et al. Body fat distribution, relative weight, and liver enzyme levels: a population based study. Hepatology. 2004; 39; 754-763 (9)
10. Fan Cy, Pan J, Usuda N, Yelandi AV, Rao MS, Reddy JK, Steatohepatitis, spontaneousperoxisome proliferation and liver tumours in mice lacking fatty acyl CoA ocidase; Implications fotedr peroxisome proliferator activated receptor alpha ligand metabolism. J Bio Chem 1998;273;15639-15645
11. Chavin KD, Yang SQ, Lin NZ et al. Obesity increases expression of uncoupling protein 2 in hepatocytes and promotes liver ATP depletion. J biol chem 1999;274:5692-5700
12. Denk H, Gschnait F, Wolff K. Hepatocellular hyalin (Mallory bodies) in long term griseofulvin-treated mice: a new experimental model for the study of hyalin formation. Lab invest 1975; 32:773-776
13. Telphy Tr, coffman BL, Ingall G et al, identification of N-methylprotophorphyran ix in livers of untreated mice and mice treated with 3,5- dieethoxycarbonyl-1.4-dihydorcollidine; source of the methyl group; Arch Bio Chem Biophys 1981; 212; 120-126
14. Purohit A, Reed MJ. Regulation of oestrogen synthesis in postmenopausal women. Steroids 2002: 67; 979-983 (9)
15. Bianchini F, Kaaka R, Vainio H. Overweight, obesity, and cancer risk. The Lancet Oncology 2002; 3: 565-574 (10)
16, Bates GW, Whitworth N. Effects of obesity on sex steroid metabolism. Journal Chronic Diseases 1982; 35: 893 (11)
17. Simpson ER. Role of aromatase in sex steroid action. Journal of Molecular Endocrinology (2000);25: 149-156 (12)
18. Hammoud AO, Gibson M, Peterson CM et al. Obesity and Male Reproductive Potential. Journal of Andrology: 27; 619 – 625 (13)
19. Lukanova A, Lundin E et al. Body mass index, circulating levels of sex steroid hormones, IGF-1 and IGF-binding protein-3: a cross-sectional study in healthy women. European Journal of Enodcrinology 2004; 190: 161-171 (14)
20. Metwally M, Li TC, Ledger L. The impact of obesity on female reproductive function. Obesity Reviews 2007; 8: 515-523 (15)
21. Bray AG. Obesity and Reproduction. Human Reproduction 1997; 12: 26-32 (16)
22. Haslam, D.W. and James, W.P Obesity. Lancet 2995; 366:1197-1209. (17)
23. Key TJ, Pike MC. The dose-effect relationship between ‘unopposed’ oestrogens and endometrial mitotic rate: its central role in explaining and predicting endometrial cancer risk. British Journal of Cancer 1988: 57; 205-12 (18)
24. Strauss JF, Dunaif A. Molecular Mysteries of Polycystic Ovary Syndrome. Molecular Endrocinology 1999: 13; 800-805 (19)
25. Rivenes, A.C., Harvey, S.B., and Mykltun, A. (2009). The relationship between abdominal fat, obesity, and common mental disorders: Results from the HUNT Study. Journal of Psychosomatic Research, 66: 269-275. (R1)
26. Stunkard, A.J., Faith, M.S. and Allison, K.C. (2003). Depression and Obesity. Society of Biological Psychiatry, 54: 330-337. (R2)
27. Mather, A.A., Brian, J.C., Enns, M.W. and Sareen, J. (2009). Association of obesity with psychiatric disorders and suicidal behaviours in a nationally representative sample. Journal of Psychosomatic Research, 66: 277-285. (R3)
28. Simon, G.E., Ludman, E.J., Linde, J.A., Operskalski, B.H., Ichikawa, L., Rohde, P., Finch, E.A. and Jeffery, R.W. (2008). Association between obesity and depression in middle-aged women. Gen Hosp Psychiatry, 30(1): 32-39. (R4)
29. Scott, K.M., Bruffaerts, R., Simon, G.E., Alonso, J., Angermeyer, M., de Girolamo, G., Demyttenaere, K., Gasquet, I., Haro, J.M., Karam, E., Kessler, R.C., Levinson, D., Mora, M.E.M., Browne, M.O., Ormel, J.H., Villa, J.P., Uda, H. and von Korff, M. (2008). Obesity and Mental Disorders in the General Population: Results from the World Mental Health Surveys. Int J Obes, 32(1):192-200. (R5)
30, Simon, G.E., Von Korff, M., Saunders, K., Miglioretti, D.L., Crane, P.K., van Belle, G. and Kessler, R.C. (2006). Association Between Obesity and Psychiatric Disorders in the US Adult Population. Arch Gen Psychiatry, 63: 824-830. (R6)
31. Carpenter, K.M., Hasin, D.S., Allison, D.B. and Faith, M.S. (2000) Relationships between obesity and DSM-IV major depressive disorder, suicide ideation, and suicide attempts: results from a general population study. Am J Public Health, 90:251–257. (R7)
32. Onyike, C.U., Crum R.M. and Lee H.B. (2003). Is obesity associated with major depression? Results from the third National Health and Nutrition Examination Survey. Am J Epidemiol, 158:1139–1147. (R8)
33. Scott, K.M., Oakley Browne, M.A., McGee, M.A. and Wells, J.E. (2006). Mental-physical comorbidity in Te Rau Hinengaro: the New Zealand Mental Health Survey (NZMHS). Australian and New Zealand Journal of Psychiatry, 40:882–888. (R9)
34. Dong, C., Li, W.D. and Li, D. (2006). Extreme obesity is associated with suicide attempts: results from a family study. Int J Obes, 30: 388-390. (R10)
35. Kleiner, K.D., Gold, M.S., Frost-Pineda, K. (2004). Body mass index and alcohol use. J Addict Dis, 23:105-118. (R11)
36. Hasler, G., Pine, D.S. and Gamma, A. (2004). The associations between psychopathology and being overweight: A 20-year prospective study. Psychol Med, 34:1047–157. (R12)
37. Faith, M.S., Matz, P.E. and Jorge, M.A. (2002). Obesity-depression associations in the population. J Psychosom Res, 53:935–942. (R13)
38. Onyike, C.U., Crum, R.M., Lee, H.B., Lyketsos, C.G. and Eaton, W.W. (2003). Is obesity associated with major depression? Results from the Third National Health and Nutrition Examination Survey. American Journal of Epidemiology. 158(12):1139–1147. (R14)
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 1.8 1.9 1. Kopelman P.G. Obesity as a medical problem. Nature 2000; 404:635-642 Cite error: Invalid
<ref>tag; name "Kopelman2000" defined multiple times with different content Cite error: Invalid<ref>tag; name "Kopelman2000" defined multiple times with different content Cite error: Invalid<ref>tag; name "Kopelman2000" defined multiple times with different content Cite error: Invalid<ref>tag; name "Kopelman2000" defined multiple times with different content Cite error: Invalid<ref>tag; name "Kopelman2000" defined multiple times with different content Cite error: Invalid<ref>tag; name "Kopelman2000" defined multiple times with different content - ↑ Boon NA, Colledge NR, Walker BR. Davidson's Principles and Practice of Medicine. 2006. p 111
- ↑ 3.0 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Gaal L.F.V. et al. Mechanisms linking obesity with cardiovascular disease. Nature 2006; 444:875-879
- ↑ 4.00 4.01 4.02 4.03 4.04 4.05 4.06 4.07 4.08 4.09 4.10 4.11 4.12 4.13 4.14 4.15 4.16 4.17 Mathew B. et al. Obesity: effects on cardiovascular disease and its diagnosis. J Am Board Fam Me 2008; 21:562-568
- ↑ 5.0 5.1 Terao S. et al. Inflammatory and injury responses to ischemic stroke in obese mice. Stroke; 2008. 39:943-950
- ↑ Boon NA, Colledge NR, Walker BR. Davidson's Principles and Practice of Medicine. 2006. p 111
- ↑ 7.0 7.1 Paul Angulo, Obesity and Nonalcoholic fatty Liver Disease, Nutrition reviews, vol 65, 6, 2007, s57-s63
- ↑ 8.0 8.1 8.2 8.3 Paul Angulo, Nonalcoholic fatty liver disease, New England Journal of Medicine, vol346, 16, 2002, 1221-123.
- ↑ Stranges S, Dorn JM, Multi P, et al. Body fat distribution, relative weight, and liver enzyme levels: a population based study. Hepatology. 2004; 39; 754-763
- ↑ Fan Cy, Pan J, Usuda N, Yelandi AV, Rao MS, Reddy JK, Steatohepatitis, spontaneousperoxisome proliferation and liver tumours in mice lacking fatty acyl CoA ocidase; Implications fotedr peroxisome proliferator activated receptor alpha ligand metabolism. J Bio Chem 1998;273;15639-15645
- ↑ Chavin KD, Yang SQ, Lin NZ et al. Obesity increases expression of uncoupling protein 2 in hepatocytes and promotes liver ATP depletion. J biol chem 1999;274:5692-5700
- ↑ 12.0 12.1 12.2 Kurt Zatloukal, conny Stumptner, Andrea Fuchsbichler, Peter Fickert, Carolin Lackner, Michael Trauner and Helmut Denk, The Keratin cytoskeleton in liver diseases, journal of pathology, 2003 vol 204: 367-376
- ↑ Denk H, Gschnait F, Wolff K. Hepatocellular hyalin (Mallory bodies) in long term griseofulvin-treated mice: a new experimental model for the study of hyalin formation. Lab invest 1975; 32:773-776
- ↑ Telphy Tr, coffman BL, Ingall G et al, identification of N-methylprotophorphyran ix in livers of untreated mice and mice treated with 3,5- dieethoxycarbonyl-1.4-dihydorcollidine; source of the methyl group; Arch Bio Chem Biophys 1981; 212; 120-126
- ↑ 15.0 15.1 15.2 Simpson ER. Role of aromatase in sex steroid action. Journal of Molecular Endocrinology (2000);25: 149-156
- ↑ Purohit A, Reed MJ. Regulation of oestrogen synthesis in postmenopausal women. Steroids 2002: 67; 979-983
- ↑ 17.0 17.1 17.2 Bianchini F, Kaaka R, Vainio H. Overweight, obesity, and cancer risk. The Lancet Oncology 2002; 3: 565-574
- ↑ 18.0 18.1 Bates GW, Whitworth N. Effects of obesity on sex steroid metabolism. Journal Chronic Diseases 1982; 35: 893
- ↑ 19.0 19.1 19.2 19.3 19.4 19.5 19.6 Hammoud AO, Gibson M, Peterson CM et al. Obesity and Male Reproductive Potential. Journal of Andrology: 27; 619 – 625
- ↑ 20.0 20.1 20.2 20.3 Lukanova A, Lundin E et al. Body mass index, circulating levels of sex steroid hormones, IGF-1 and IGF-binding protein-3: a cross-sectional study in healthy women. European Journal of Enodcrinology 2004; 190: 161-171
- ↑ Rivenes, A.C., Harvey, S.B., and Mykltun, A. (2009). The relationship between abdominal fat, obesity, and common mental disorders: Results from the HUNT Study. Journal of Psychosomatic Research, 66: 269-275
- ↑ Stunkard, A.J., Faith, M.S. and Allison, K.C. (2003). Depression and Obesity. Society of Biological Psychiatry, 54: 330-337.
- ↑ Rivenes, A.C., Harvey, S.B., and Mykltun, A. (2009). The relationship between abdominal fat, obesity, and common mental disorders: Results from the HUNT Study. Journal of Psychosomatic Research, 66: 269-275
- ↑ Stunkard, A.J., Faith, M.S. and Allison, K.C. (2003). Depression and Obesity. Society of Biological Psychiatry, 54: 330-337.