Leucine zipper
Jump to navigation
Jump to search
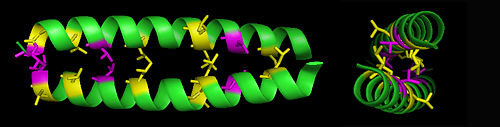
Leucine zippers are a commonly occuring structural motif in protein structures, particularly in DNA-binding proteins, in which the amino acid leucine is repeated every seven amino acids within an alpha-helix structure. Additional leucines or valines may be present every 3rd or 4th position between the leucines. This sequence of amino acids creates an -helix with a very hydrophobic face, so that two such proteins can form what is termed a coiled-coil structure. Both homodimer and heterodimer leucine zippers occur naturally.
References
- ↑ E.K. O'Shea,J.D.Klemm,P.S.Kim and T.Alber (1991). "X-Ray Structure of the GCN4 Leucine Zipper, a Two-Stranded Coiled Coil". Science 254: 539.