User:Milton Beychok/Sandbox
Measurement of density
For a homogeneous solid object, the mass divided by the volume gives the density. The mass is normally measured with an appropriate weighing scale or balance and the volume may be measured directly (from the geometry of the object) or by the displacement of a liquid.
A very simple and commonly used instrument for directly measuring the density of a liquid is the hydrometer. A less common device for measuring fluid density is a pycnometer and a similar device for measuring the absolute density of a solid is a gas pycnometer.
Another instrument used to determine the density of a liquid or a gas is the digital density meter, based on the oscillating U-tube principle.
Density of irregular shaped solid object heavier than water
One of the many statements attributed to the Archimedes principle is that an object immersed in a fluid apparently loses weight by an amount equal to the weight of the fluid displaced.[1][2][3] That principle makes it possible to determine of the density of a solid object that is denser than water and is so irregular in shape that its volume cannot be measured directly.
First, the object's dry weight in air, , is measured. Next, the weight of the object while submerged in water, , is measured. Then the submerged object is removed from the water, the excess surface water is removed and the wet weight in air, , is measured.[4] The density of the object, , is then obtained from:
and the volumetric porosity fraction, can be obtained from:
Density of solid objects by x-ray diffraction crytallography
For some crystalline materials, density can be calculated using this equation which is based the molecular mass of the material and on information provided by x-ray crystallography:
where:
- M is molecular mass
- L is Loschmidt or Avogadro's constant
- a, b, c are the lattice parameters
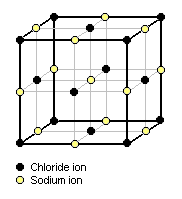
For example, the cubic lattice structure of crystalline sodium chloride (NaCl) is depicted in Figure 1.[5] Each sodium ion is surrounded by six chloride ions and each chloride ion is surrounded by six sodium ions. The sodium chloride molecule, as such, has disappeared. This structural arrangement is in conformity with the x-ray reflection spectra.
From a study of Figure 1, it can be deduced that each corner chloride ion is common to eight adjacent cubic cells so that only one-eighth of its mass contributes to the mass of the unit cell and thus all eight corner chloride units contribute the mass of one chloride ion. Likewise, the centered chloride ion on each face of the cubic cell is common to two adjacent cubic cells and thus all six of the face-centered chloride ions contribute the mass of three chloride ions. The total mass of the chlorine in the cell is therefore equal to the mass of four chloride ions.
Similarly, it can be deduced that each of the twelve sodium ions on the external faces of the cubic cell is common to four adjacent cubic cells so that only one-fourth of its mass contributes to the mass of the unit cell and thus the twelve external face sodium ions contribute the mass of three sodium ions. The one sodium ion in the center of the cell contributes all of it mass to the mass of the unit cell. The total mass of sodium in the cell is therefore equal to the mass of four sodium ions, and total mass of the cell may be taken as the mass of four molecules of sodium chloride.
The molecular mass of sodium chloride is 58.54 g/mol and the cube's lattice parameters a, b and c (height, width and length) cell are each 0.563 nm.
- ↑ California State University Dominguez Hills
- ↑ Murat Bengisu. Engineering Ceramics, First Edition. Springer, pages 363-366. ISBN 3-540-67687-2.
- ↑ ASTM C373-88 Standard Test Method for Water Absorption, Bulk Density, Apparent Porosity and Apparent Specific Gravity of Fired Whiteware
- ↑ American Society of Composites (1994). American Society of Composites, Ninth International Conference Proceedings. CRC Press, page 539. 1-56676-220-0.
- ↑ Alexander Findlay (1942). Practical Physical Density. Longman, Greens and Company, 164-167.