Cephalosporin: Difference between revisions
imported>David E. Volk No edit summary |
mNo edit summary |
||
| (13 intermediate revisions by 4 users not shown) | |||
| Line 1: | Line 1: | ||
{{subpages}} | {{subpages}} | ||
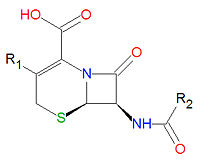
{{Image|Cephalosporin base.jpg|right|200px|Base structure of all cephalosporins.}} | |||
''' | '''Cephalosporins''' are a class of [[antibiotic]] compounds sharing a common base structure, 7-aminocephalosporanic acid (7-ACA), that was derived from the first cephalosporin discovered, [[cephalosporin C]]. [[Penicillin]]s are very similar, although they contain a five-membered ring in place of the six-membered ring present in the cephalosporin. The activity of cephalosporins, penicillins, and some other antibiotics are due to the presence of a [[lactam|beta-lactam]], which binds irreversibly, via acylation, to penicillin-binding proteins, thereby inhibiting the peptidogycan layer of bacterial cell wall synthesis. Cephalosporins are often made semisynthetically. Cephalosporins and the very closely related[[cephamycin]]s are collectively referred to as [[cephems]]. | ||
== Nomenclature == | |||
Because the original cephalosporins used the "ceph" form of the spelling and were often trademarked, the [[International Nonproprietary Name]]s (INN) suggested by the [[World Health Organization]] use the "cef" spelling for the generic drug name of all cephalosporins. | |||
== First generation cephalosporins == | == First generation cephalosporins == | ||
| Line 34: | Line 37: | ||
* [[Cefotetan]] | * [[Cefotetan]] | ||
* [[Cefoxitin]] | * [[Cefoxitin]] | ||
== Third generation cephalosporins == | |||
The third generation has additional activity against ''[[Pseudomonas aeruginosa]]''. | |||
* [[Cefcapene]] | |||
* [[Cefdaloxime]] | |||
* [[Cefdinir]] | |||
* [[Cefditoren]] | |||
* [[Cefetamet]] | |||
* [[Cefixime]] | |||
* [[Cefmenoxime]] | |||
* [[Cefodizime]] | |||
* [[Cefoperazone]] | |||
* [[Cefotaxime]] | |||
* [[Cefpimizole]] | |||
* [[Cefpodoxime]] | |||
* [[Ceftazidime]] | |||
* [[Cefteram]] | |||
* [[Ceftibuten]] | |||
* [[Ceftiofur]] | |||
* [[Ceftiolene]] | |||
* [[Ceftizoxime]] | |||
* [[Ceftriaxone]] | |||
== Fourth generation cephalosporins== | |||
While still active against ''[[Enterobacteriaceae]]'' and ''[[Pseudomonas aeruginosa]]'', this class has much better activity against gram-positive organisms than the third-generation antipseudomonals. The single approved agent is less effective against [[methicillin-resistant Staphylococcus aureus]] than the first and second generations. It is not effective against ''[[Bacteroides fragilis]]''. | |||
* [[Cefepime]][[Category:Suggestion Bot Tag]] | |||
Latest revision as of 11:01, 26 July 2024
Cephalosporins are a class of antibiotic compounds sharing a common base structure, 7-aminocephalosporanic acid (7-ACA), that was derived from the first cephalosporin discovered, cephalosporin C. Penicillins are very similar, although they contain a five-membered ring in place of the six-membered ring present in the cephalosporin. The activity of cephalosporins, penicillins, and some other antibiotics are due to the presence of a beta-lactam, which binds irreversibly, via acylation, to penicillin-binding proteins, thereby inhibiting the peptidogycan layer of bacterial cell wall synthesis. Cephalosporins are often made semisynthetically. Cephalosporins and the very closely relatedcephamycins are collectively referred to as cephems.
Nomenclature
Because the original cephalosporins used the "ceph" form of the spelling and were often trademarked, the International Nonproprietary Names (INN) suggested by the World Health Organization use the "cef" spelling for the generic drug name of all cephalosporins.
First generation cephalosporins
- Cefacetrile
- Cefadroxil
- Cefalexin
- Cefaloglycin
- Cefalonium
- Cefaloridine
- Cefalotin
- Cefapirin
- Cefatrizine
- Cefazaflur
- Cefazedone
- Cefazolin
- Cefradine
- Cefroxadine
- Ceftezole
Second generation cephalosporins
In general, second generation cephalosporins have a broader spectrum of activity against Gram-negative bacteria.
Third generation cephalosporins
The third generation has additional activity against Pseudomonas aeruginosa.
- Cefcapene
- Cefdaloxime
- Cefdinir
- Cefditoren
- Cefetamet
- Cefixime
- Cefmenoxime
- Cefodizime
- Cefoperazone
- Cefotaxime
- Cefpimizole
- Cefpodoxime
- Ceftazidime
- Cefteram
- Ceftibuten
- Ceftiofur
- Ceftiolene
- Ceftizoxime
- Ceftriaxone
Fourth generation cephalosporins
While still active against Enterobacteriaceae and Pseudomonas aeruginosa, this class has much better activity against gram-positive organisms than the third-generation antipseudomonals. The single approved agent is less effective against methicillin-resistant Staphylococcus aureus than the first and second generations. It is not effective against Bacteroides fragilis.