Steroid: Difference between revisions
imported>Gareth Leng No edit summary |
imported>Gareth Leng No edit summary |
||
| Line 2: | Line 2: | ||
{{TOC-right}} | {{TOC-right}} | ||
'''Steroids''', or '''steroid hormones''', are powerful [[hormone]]s with drastic effects, both good and bad, when artificially supplemented into living systems. They are normally produced by three glands, the [[adrenal cortex]], the [[testes]], and the [[ovaries]], but are also produced by the [[placenta]] during pregnancy, and some steroids ("neurosteroids") are produced within the brain. The five major classes of steroids are: [[progestagen]]s, [[glucocorticoids]], [[mineralcorticoid]]s, [[androgen]]s and [[estrogen]]s. Steroids play an important role at all stages of life from the embryo until death. [[Corticosteroids]], or synthetic mimics such as [[prednisone]], are used to treat inflammation related illnesses like asthma or rheumatoid arthritis, but they can have severe side effects. Athletes have often taken anabolic steroids to improve muscle growth and athletic performance. Glucocorticoids play a role in metabolism and inflammation, and estrogens have been linked to cancer. [[Testosterone]] and estrogen influence sexual traits (maleness/femaleness). All steroid hormones are naturally synthesized from [[cholesterol]] (through [[pregnenolone]]) under the control of the [[anterior pituitary gland]], which produces [[andrenocorticotropic hormone]] (ACTH, or corticotropin), a polypeptide that stimulates the conversion of cholesterol to pregnenolone. The steroid hormones activate gene expression by binding to enhancer proteins, called [[steroid receptors]]. Deficiency of the enzyme [[21-hydroxylase]] is the most common steroid-related in-born metabolic disorder, but treatments are available for this condition. | '''Steroids''', or '''steroid hormones''', are powerful [[hormone]]s with drastic effects, both good and bad, when artificially supplemented into living systems. They are normally produced by three glands, the [[adrenal cortex]], the [[testes]], and the [[ovaries]], but are also produced by the [[placenta]] during pregnancy, and some steroids ("neurosteroids") are produced within the brain. The five major classes of steroids are: [[progestagen]]s, [[glucocorticoids]], [[mineralcorticoid]]s, [[androgen]]s and [[estrogen]]s. Steroids play an important role at all stages of life from the embryo until death. [[Corticosteroids]], or synthetic mimics such as [[prednisone]], are used to treat inflammation related illnesses like [[asthma]] or [[rheumatoid arthritis]], but they can have severe side effects. Athletes have often taken [[anabolic steroids]] to improve muscle growth and athletic performance. Glucocorticoids play a role in metabolism and inflammation, and estrogens have been linked to cancer. [[Testosterone]] and estrogen influence sexual traits (maleness/femaleness). All steroid hormones are naturally synthesized from [[cholesterol]] (through [[pregnenolone]]) under the control of the [[anterior pituitary gland]], which produces [[andrenocorticotropic hormone]] (ACTH, or corticotropin), a polypeptide that stimulates the conversion of cholesterol to pregnenolone. The steroid hormones activate gene expression by binding to enhancer proteins, called [[steroid receptors]]. Deficiency of the enzyme [[21-hydroxylase]] is the most common steroid-related in-born metabolic disorder, but treatments are available for this condition. | ||
==Classification and numbering == | ==Classification and numbering == | ||
Steroids can be classified into five major groups, based on their structures and functions:progestagens, glucocorticoids, mineralcorticoids, androgens and estrogens. The glucocorticoids and mineralcorticoids are referred to collectively as the corticosteroids, and the androgens and estrogens are referred to collectively as sex steroids. The | Steroids can be classified into five major groups, based on their structures and functions: progestagens, ''glucocorticoids, mineralcorticoids, androgens and estrogens''. The glucocorticoids and mineralcorticoids are referred to collectively as the 'corticosteroids', and the androgens and estrogens are referred to collectively as 'sex steroids' or 'gonadal steroids'. The progestagens are vital to the start of life, via pregnancy, while the glucocortioids and mineralocorticoids are vital to metabolism and electrolyte balance. The sex steroids provide the [[sexual dimorphism]] observed in many species. Because all steroids are derived from cholesterol, the numbering and nomenclature follow that of [[cholesterol]]. | ||
===Progestagens=== | ===Progestagens=== | ||
| Line 13: | Line 13: | ||
[[Image:Progesterone2.jpg|left|thumb|150px]] | [[Image:Progesterone2.jpg|left|thumb|150px]] | ||
Progesterone, a [[progestagen]] which prepares the lining of the uterus for implantation of an ovum, is biosynthesized from prognenolone by oxidation of the 3-hydroxy group into a 3-keto group and by the [[isomerization]] of the <math>\delta</math><sup>5</sup> double bond into a <math>\delta</math><sup>4</sup> double bond. This hormone is also essential to maintain pregnancy. Progesterone is the precursor chemical in the biosynthesis of corticoids and androgens. | Progesterone, a [[progestagen]] which prepares the lining of the uterus for implantation of an ovum, is biosynthesized from prognenolone by oxidation of the 3-hydroxy group into a 3-keto group and by the [[isomerization]] of the <math>\delta</math><sup>5</sup> double bond into a <math>\delta</math><sup>4</sup> double bond. This hormone is also essential in mammals to maintain pregnancy. Progesterone is the precursor chemical in the biosynthesis of corticoids and androgens. | ||
==Biochemistry== | ==Biochemistry== | ||
| Line 33: | Line 33: | ||
===Sex steroids=== | ===Sex steroids=== | ||
[[Image:Androgen estrogen synthesis structures.jpg|right|thumb|400px|{{#ifexist:Template:Androgen estrogen synthesis structures.jpg/credit|{{Androgen estrogen synthesis structures.jpg/credit}}<br/>|}}]] | [[Image:Androgen estrogen synthesis structures.jpg|right|thumb|400px|{{#ifexist:Template:Androgen estrogen synthesis structures.jpg/credit|{{Androgen estrogen synthesis structures.jpg/credit}}<br/>|}}]] | ||
The term sex steroids refers collectively to the androgens and the estrogens, which both play a major role in defining sexual traits. Disorders affecting sex steroid levels can lead to the masculinization of women and the demasculinization of men. | The term sex steroids refers collectively to the androgens and the estrogens, which both play a major role in defining sexual traits. Disorders affecting sex steroid levels can lead to the masculinization of women and the demasculinization (feminization) of men. | ||
====Biochemistry==== | ====Biochemistry==== | ||
| Line 41: | Line 41: | ||
====Medical use of androgens==== | ====Medical use of androgens==== | ||
Testosterone supplementation of elderly males with low serum testosterone levels has mixed results.<ref name="pmid18167405">{{cite journal |author=Emmelot-Vonk MH | Testosterone supplementation of elderly males with low serum testosterone levels has mixed results.<ref name="pmid18167405">{{cite journal |author=Emmelot-Vonk MH ''et al'' |title=Effect of testosterone supplementation on functional mobility, cognition, and other parameters in older men: a randomized controlled trial |journal=JAMA |volume=299 |pages=39–52 |year=2008 |pmid=18167405 |doi=10.1001/jama.2007.51}}</ref> | ||
====Medical use of estrogens==== | ====Medical use of estrogens==== | ||
| Line 65: | Line 65: | ||
;Manifestations | ;Manifestations | ||
Aldosterone is the main mineralcorticoid and is produced in the adrenal glands. It plays a critical role in | Aldosterone is the main mineralcorticoid and is produced in the adrenal glands. It plays a critical role in maintaining normal plasma concentrations of sodium; patients with 21-hydroxylase deficiency can suffer hypertension and dehydration, which can lead to shock or death. Increases in the levels of the androgens (testosterone, for example) resulting from the overproduction of progesterone and 17<math>\alpha</math>-hydroxy-progesterone lead to the [[virilization]] ((masculinization) of those affected. In girls, masculinization of the external genitalia is usually evident at birth. Boys appear normal at birth but sexual precocity soon follows. Accelerated growth and early bone development leads to a shortened stature. | ||
==References== | ==References== | ||
Revision as of 08:42, 14 January 2009
Template:TOC-right Steroids, or steroid hormones, are powerful hormones with drastic effects, both good and bad, when artificially supplemented into living systems. They are normally produced by three glands, the adrenal cortex, the testes, and the ovaries, but are also produced by the placenta during pregnancy, and some steroids ("neurosteroids") are produced within the brain. The five major classes of steroids are: progestagens, glucocorticoids, mineralcorticoids, androgens and estrogens. Steroids play an important role at all stages of life from the embryo until death. Corticosteroids, or synthetic mimics such as prednisone, are used to treat inflammation related illnesses like asthma or rheumatoid arthritis, but they can have severe side effects. Athletes have often taken anabolic steroids to improve muscle growth and athletic performance. Glucocorticoids play a role in metabolism and inflammation, and estrogens have been linked to cancer. Testosterone and estrogen influence sexual traits (maleness/femaleness). All steroid hormones are naturally synthesized from cholesterol (through pregnenolone) under the control of the anterior pituitary gland, which produces andrenocorticotropic hormone (ACTH, or corticotropin), a polypeptide that stimulates the conversion of cholesterol to pregnenolone. The steroid hormones activate gene expression by binding to enhancer proteins, called steroid receptors. Deficiency of the enzyme 21-hydroxylase is the most common steroid-related in-born metabolic disorder, but treatments are available for this condition.
Classification and numbering
Steroids can be classified into five major groups, based on their structures and functions: progestagens, glucocorticoids, mineralcorticoids, androgens and estrogens. The glucocorticoids and mineralcorticoids are referred to collectively as the 'corticosteroids', and the androgens and estrogens are referred to collectively as 'sex steroids' or 'gonadal steroids'. The progestagens are vital to the start of life, via pregnancy, while the glucocortioids and mineralocorticoids are vital to metabolism and electrolyte balance. The sex steroids provide the sexual dimorphism observed in many species. Because all steroids are derived from cholesterol, the numbering and nomenclature follow that of cholesterol.
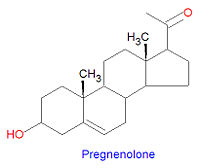
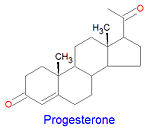
Progestagens
Pregnenolone is the first steroid derived from cholesterol. It is synthesized through an intermediate, 20,22-dihydroxy-cholesterol, which is subsequently oxidized at C-20 to form a ketone with cleavage of carbons 22-27.
Progesterone, a progestagen which prepares the lining of the uterus for implantation of an ovum, is biosynthesized from prognenolone by oxidation of the 3-hydroxy group into a 3-keto group and by the isomerization of the 5 double bond into a 4 double bond. This hormone is also essential in mammals to maintain pregnancy. Progesterone is the precursor chemical in the biosynthesis of corticoids and androgens.
Biochemistry
Synthesis
Cholesterol is the precursor from which all steroid hormones are synthesized. All steroid hormones have 21 or fewer carbons, although their precursor cholesterol has 27 carbon atoms. Because of this, all steroid numbering and nomenclature follow that of cholesterol. Some cholesterol derivatives have a proton added to the C-5 carbon. If the H-5 proton is -oriented (points down) then rings A and B are fused in a trans conformation, but in the orientation, the rings are fused in a cis conformation. All steroids with an H5 are in the 5 orientation, while bile salts derived from cholesterol have the opposite 5 orientation.
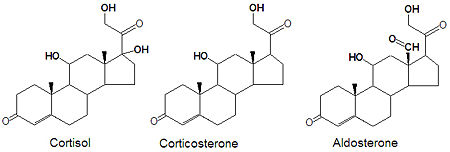
Corticosteroids
Biochemistry
Glucocorticoids and mineralocorticoids (or mineralcorticoids) are naturally synthesized by the enzymatic oxidation of progesterone. Cortisol, the major glucocorticoid, is produced when progesterone is hydroxylated at three positions, C-11, C-17 and C21. The oxidation of the C-17 carbon must occur before the hydroxylation at C-21 to synthesize cortisol, otherwise corticosterone is formed. Aldosterone, the major mineralcorticoid, is synthesized from corticosterone by oxidation of the C-18 methyl group to form an aldehyde.
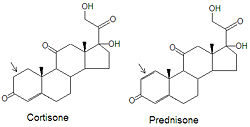
Medical use of glucocorticoids
Although cortisone can be used to treat inflammation related diseases, like rheumatoid arthritis, it has severe side effects. The drug prednisone and other glucocorticoids have been developed as a cortisone mimic and its use leads to milder side effect problems in the patient population. The only difference between the two compounds is the presence of an double bond between the C1 and C2 carbons of prednisone (note arrows in figure). Cortisone has a single C-C bond in this location.
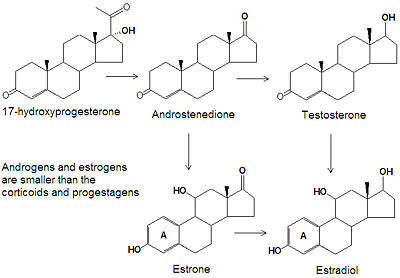
Sex steroids
The term sex steroids refers collectively to the androgens and the estrogens, which both play a major role in defining sexual traits. Disorders affecting sex steroid levels can lead to the masculinization of women and the demasculinization (feminization) of men.
Biochemistry
Androgens and estrogens have only nineteen or eighteen carbon atoms, respectively, compared to the larger progestagens, glucocorticoids and mineralcorticoids all have twenty one carbon atoms. The biosynthesis of both classes of sex steroids starts with the hydroxylation of progesterone at C-17 to produce 17-hydroxyprogesterone. Cleavage of the C20-C21 side chain and oxidation of the C-17 hydroxyl group into a ketone produces androstenedione. Reduction of the C-17 keto group of androstenedione yields testosterone, one of the most abused anabolic steroids.
The smallest class of steroids, the estrogens, are biosynthesized from the androgens by loss of the C-19 -methyl group, reduction of the C-3 ketone to a hydroxyl group, and the formation of an aromatic ring A. Estradiol is the major estrogen.
Medical use of androgens
Testosterone supplementation of elderly males with low serum testosterone levels has mixed results.[1]
Medical use of estrogens
See estrogen and contraception (medical methods).
Abuse of anabolic steroids
Anabolic steroids are steroids related to testosterone which encourage muscle development. They work by increasing the production of proteins and increase cell tissue, especially muscle mass. In addition, they decrease muscle catabolism by lowering the levels of cortisol, a stress hormone. Anabolic steroids have other effects, including virilization, resulting in larger clitoris in women and penis in men, hair growth, acne, elevated blood pressure, liver and heart damage, and increases in LDL cholesterol and lower HDL cholesterol levels. The use of anabolic steroids is banned in many sports and their use without valid prescriptions is illegal in many countries.
Mechanism of action
Classically, steroids exert their effects by "genomic" actions - by altering gene expression in their target cells. Because of this mode of action, steroid hormones take hours to have an effect because messenger RNA and then proteins must be synthesized; this is in contrast with other fast acting hormones, such as epinephrine, which have immediate effects on their target cells. The genomic actions of steroids are mediated by specific receptors that are located in the cytosol of particular cells. Steroids enter cells freely (they are lipid soluble and so can penetrate cell membranes); if a cell expresses specific receptors for that steroid. The formation of the messenger RNA can be suppressed by actinomycin D. Steroids first bind tightly, with dissociation constants in the nanomolar range, to their receptor protein. This complex is tranlocated into the cell nucleus and binds to specific operator sites in the DNA. All steroid receptors have a DNA-binding region, often called the DNA-binding domain (or DBD), which is rich is the positively-charged amino acids arginine and lysine, and also contains many cysteines. The presence of a "Cys-X-X-Cys" motif (X=any amino acid) suggests the presence of a metal-binding finger, however, no structures of steroid receptors in the unbound state have ever been determined to verify this. The receptors also have activation factor domains at which additional cofactors must bind before the steroid:receptor complex can enter the nucleus or inititiate gene expression. Due to their complexity, hormone receptors are not well understood at this time and are the subject of intense research. Several steroid:receptor complex structures have recently been determined. In addition to the DBD, all steroid hormone receptors have a hormone-binding region and the similarities in this region of the receptors suggests that they all had a common ancestor. The steroid receptor proteins are generally specific for each steroid class, binding tightly only to the target steroids, but weak binding can occur with other steroids for some of them. The thyroid-hormone receptor is similar in structure to the steroid receptors.
In addition to these classic genomic actions, some steroids also have direct effects on cell membranes which mediate immediate effects. Some of these are mediated by membrane-bound steroid receptors, others are mediated by interactions of steroids with other membrane-bound proteins. for example, in the brain, progesterone is metabolised into allopregnanolone which interacts with some classaes of receptors for the inhibitory neurotransmitter GABA.
Diseases of steroid metabolism
Adrenal insufficiency
Congenital Adrenal Hyperplasia
Congenital adrenal hyperplasia is a "group of inherited disorders of the adrenal glands, caused by enzyme defects in the synthesis of cortisol (hydrocortisone) and/or aldosterone leading to accumulation of precursors for androgens. Depending on the hormone imbalance, congenital adrenal hyperplasia can be classified as salt-wasting, hypertensive, virilizing, or feminizing. The most common defect is in steroid 21-hydroxylase. Other defects occur in steroid 11-beta-hydroxylase, steroid 17-alpha-hydroxylase, or 3-beta-hydroxysteroid dehydrogenase (3-hydroxysteroid dehydrogenases)".[2] [3][4]
- Biochemistry
Some people inherit defective forms of the enzyme 21-hydroxylase. Because 21-hydroxylase is a key enzyme in the production of the corticosteroids, defects in this enzyme lead to severe physiological effects. The enzyme is required for the production of cortisol, corticosterone, aldosterone and other steroids. Diminished levels of the glucocorticoids induces the anteterior pituitary gland to increase production of ACTH, which in turn increases the levels of progesterone and 17hydroxy-progesterone. High levels of these two hormones increases the levels of androgens, which are derived from them. The pathogenesis can be stopped by the administration of glucocorticoids to complete the feed-back mechanism and halt overproduction of ACTH.
- Manifestations
Aldosterone is the main mineralcorticoid and is produced in the adrenal glands. It plays a critical role in maintaining normal plasma concentrations of sodium; patients with 21-hydroxylase deficiency can suffer hypertension and dehydration, which can lead to shock or death. Increases in the levels of the androgens (testosterone, for example) resulting from the overproduction of progesterone and 17-hydroxy-progesterone lead to the virilization ((masculinization) of those affected. In girls, masculinization of the external genitalia is usually evident at birth. Boys appear normal at birth but sexual precocity soon follows. Accelerated growth and early bone development leads to a shortened stature.
References
- ↑ Emmelot-Vonk MH et al (2008). "Effect of testosterone supplementation on functional mobility, cognition, and other parameters in older men: a randomized controlled trial". JAMA 299: 39–52. DOI:10.1001/jama.2007.51. PMID 18167405. Research Blogging.
- ↑ National Library of Medicine. Adrenal Hyperplasia, Congenital. Retrieved on 2008-01-01.
- ↑ Merke DP, Bornstein SR (2005). "Congenital adrenal hyperplasia". Lancet 365 (9477): 2125–36. DOI:10.1016/S0140-6736(05)66736-0. PMID 15964450. Research Blogging.
- ↑ Speiser PW, White PC (2003). "Congenital adrenal hyperplasia". N. Engl. J. Med. 349 (8): 776–88. DOI:10.1056/NEJMra021561. PMID 12930931. Research Blogging.